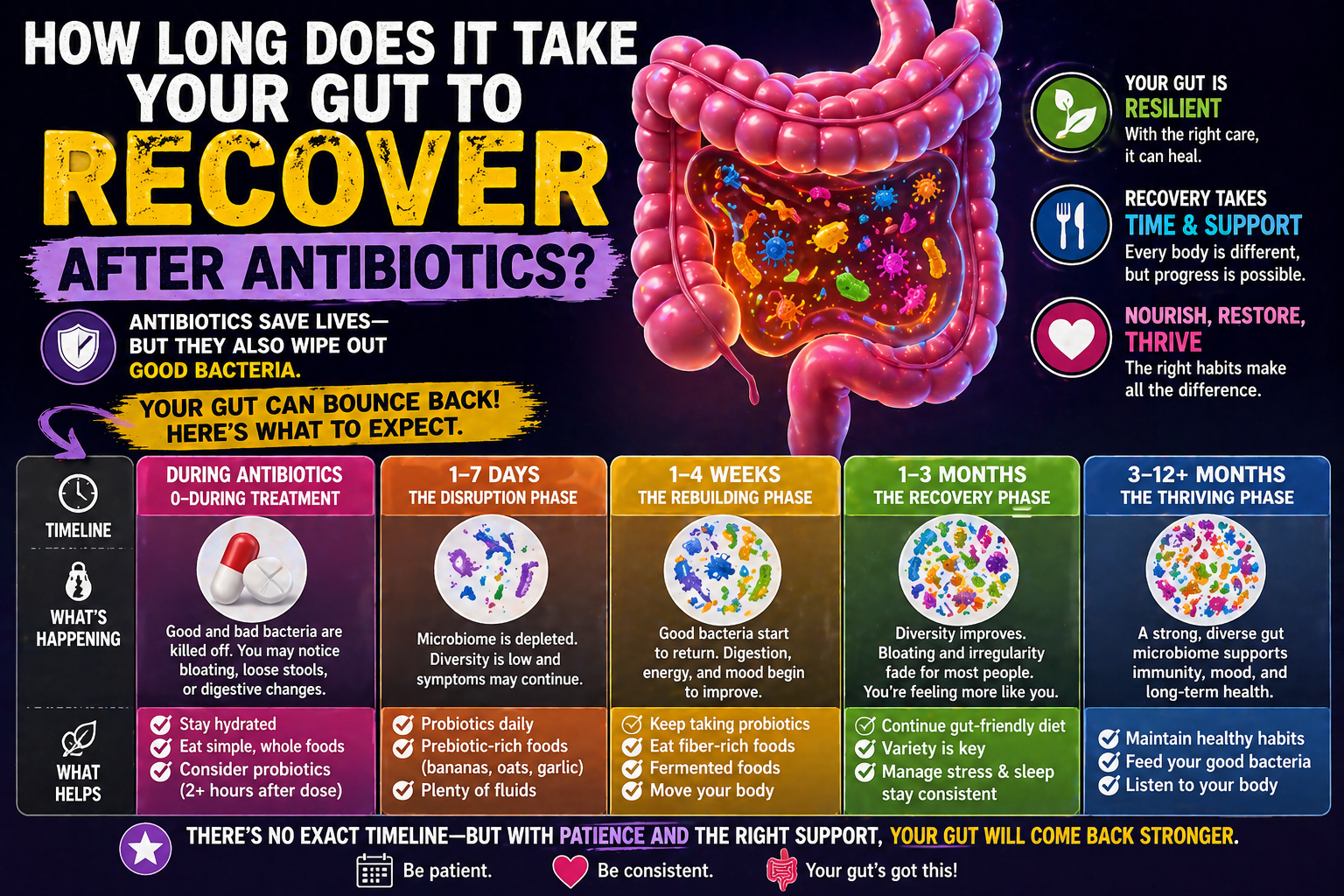
If your gut was never the same after a course of antibiotics, it may not be a coincidence. Here’s the documented link between antibiotic use and IBS — and what actually reverses it.
If Your Gut Was Never the Same After a Course of Antibiotics, This Might Explain Why
You remember when it started. Maybe it was the antibiotics you took for a chest infection two years ago. Maybe it was a round of ciprofloxacin after a UTI. Or the clindamycin prescribed for a dental procedure. You finished the course, the infection cleared, and then — your gut never quite went back to normal.
The bloating that appeared from nowhere. The unpredictable bowel habits. The cramping that shows up without warning. The foods that you used to eat without thinking twice that now reliably cause problems. You’ve been told you have IBS. But nobody mentioned that antibiotics could have been the trigger — and the research suggests that for a significant number of IBS sufferers, they absolutely were.
If antibiotics triggered your IBS — your gut can still recover
Seed DS-01 Daily Synbiotic — 24 clinically studied strains, acid-resistant delivery, prebiotic included. The most comprehensive probiotic for post-antibiotic IBS recovery. Start now and commit to 90 days.
👉 Check the current price of Seed DS-01 on Amazon
What Post-Infectious IBS Actually Is
Post-infectious IBS — PI-IBS — is a clinically recognized condition where IBS develops following a gastrointestinal infection or significant gut disruption. It’s been studied for decades in the context of food poisoning and gastroenteritis, and the research on antibiotic-triggered PI-IBS has been growing significantly over the past ten years.
The mechanism is not mysterious. Antibiotics disrupt the gut microbiome significantly — reducing bacterial diversity by 25 to 50% in the space of a week. This disruption creates measurable changes in gut barrier function, gut nerve sensitivity, gut motility regulation, and immune function in the gut wall. When these changes persist beyond the acute disruption period — which they do in a significant proportion of people — they produce the exact constellation of symptoms that define IBS: altered bowel habits, abdominal pain, bloating, and visceral hypersensitivity.
The distinction between PI-IBS and other forms of IBS matters clinically because the underlying mechanism — microbiome disruption driving gut dysfunction — is directly addressable. It’s not a permanent structural change. It’s a functional disruption that responds to the right intervention.
What the Research Shows
The evidence linking antibiotic use to subsequent IBS development is substantial and growing.
A large population-based study published in the journal Gut found that people who took antibiotics had a significantly higher risk of developing IBS in the following year compared to those who didn’t — with the risk increasing with each additional antibiotic course. The effect was strongest for broad-spectrum antibiotics and for courses taken in adulthood rather than childhood.
A 2019 meta-analysis looking at post-infectious IBS across multiple infection types found that antibiotic use was consistently associated with higher rates of subsequent IBS development — independent of the infection being treated. This is a critical finding — it suggests the antibiotic itself, not just the infection, contributes to IBS risk.
Research on the microbiome of IBS patients consistently shows measurably different gut bacterial composition compared to healthy controls — less Bifidobacterium, less Faecalibacterium prausnitzii (a key butyrate producer and anti-inflammatory species), and more gas-producing harmful species. This pattern is identical to the microbiome disruption pattern that antibiotics produce — providing a plausible, specific biological mechanism linking antibiotic exposure to IBS development.
The research isn’t suggesting that antibiotics always cause IBS — they don’t. But for people with a susceptible gut, a disrupted microbiome baseline, or significant antibiotic exposure, the connection is real, documented, and mechanistically coherent.
Why Some People Develop IBS After Antibiotics and Others Don’t
This is the question that matters for understanding your own situation. Not everyone who takes antibiotics develops IBS — so what determines who does?
Baseline microbiome diversity. People with high microbiome diversity before antibiotic exposure have more bacterial redundancy — when some species are depleted, others compensate. People with lower baseline diversity have less buffer and are more vulnerable to functional disruption from the same antibiotic exposure.
Which antibiotic was taken. Broad-spectrum antibiotics — ciprofloxacin, clindamycin, metronidazole — produce more extensive microbiome disruption than narrow-spectrum agents. Clindamycin in particular is associated with the most significant and persistent gut microbiome disruption and the highest subsequent IBS risk in population studies.
Number of courses. A single antibiotic course produces a specific level of disruption that most microbiomes can recover from with active support. Multiple courses — particularly within a short period — produce cumulative disruption that takes significantly longer to recover from and carries higher IBS risk.
Whether active recovery support was provided. People who take a quality probiotic during and after antibiotic courses, eat fermented foods, and maintain high-fiber diets show faster and more complete microbiome recovery — and presumably lower risk of the persistent disruption that drives PI-IBS.
Genetic and immunological factors. Some people appear genetically predisposed to visceral hypersensitivity and altered gut-brain axis signaling — the mechanisms that underlie IBS. For these individuals, a significant gut disruption event like an antibiotic course may be sufficient to shift a subclinical predisposition into a symptomatic condition.
How Antibiotics Trigger the Specific Mechanisms of IBS
Understanding exactly how antibiotic-induced microbiome disruption produces IBS symptoms explains why targeted gut health intervention is the most logical treatment approach.
Visceral hypersensitivity. Beneficial gut bacteria produce compounds that regulate the sensitivity of gut nerve endings — including serotonin-modulating metabolites and short-chain fatty acids that maintain normal nerve signaling thresholds. When these bacteria are depleted, gut nerve sensitivity increases. Normal levels of gas, food, and intestinal contractions that were previously imperceptible register as pain and discomfort. This heightened sensitivity is one of the defining features of IBS and it has a direct bacterial mechanism that’s addressable through microbiome restoration. Why gut pain is inconsistent and threshold-driven explains visceral hypersensitivity in more accessible detail.
Altered gut motility. Bifidobacterium and Lactobacillus species produce compounds that regulate the muscular contractions of the gut wall — the peristaltic waves that move food through at the right speed. When these populations are depleted by antibiotics, motility regulation is impaired. For some people this tips toward constipation-dominant patterns. For others — particularly those with a more reactive gastrocolic reflex — it tips toward diarrhea-dominant patterns. For many it produces the alternating, unpredictable pattern that characterizes IBS-M. The right probiotic for your specific IBS type addresses the motility component directly.
Increased gut permeability. Antibiotic-depleted gut bacteria produce less butyrate — the primary fuel for colonocytes and the main compound that maintains tight junction integrity. With less butyrate, the tight junctions between intestinal cells loosen, increasing gut permeability. This allows bacterial components and partially digested food proteins to cross into the bloodstream, triggering the systemic immune response that underlies the chronic low-grade gut inflammation characteristic of IBS. How leaky gut drives systemic symptoms covers this mechanism in detail — including the anxiety and brain fog that often accompany antibiotic-triggered IBS.
Dysbiosis-driven fermentation. With harmful gas-producing bacteria having proliferated in the ecological space antibiotics cleared, even normal food intake produces excessive fermentation — more gas, more bloating, more abdominal pressure than the same food produced before antibiotic exposure. Why bloating happens after every meal maps directly to this post-antibiotic fermentation disruption.
Can Antibiotic-Triggered IBS Be Reversed?
This is the most important question — and the answer is more hopeful than most people with post-antibiotic IBS have been led to believe.
The functional disruptions that antibiotics produce — visceral hypersensitivity, altered motility, increased permeability, dysbiotic fermentation — are not permanent structural changes. They’re functional consequences of a disrupted microbiome, and the microbiome can be deliberately restored with the right consistent intervention.
Research on probiotic treatment for PI-IBS consistently shows improvement in symptoms — including reduction in visceral hypersensitivity scores, improved motility regulation, and reduced bloating — with 8 to 16 weeks of consistent multi-strain probiotic use. The effect size is larger for PI-IBS than for IBS without an identifiable infectious or antibiotic trigger — because the mechanism is more specifically bacterial and therefore more directly addressable by bacterial intervention.
The timeline is longer than for acute post-antibiotic recovery — if antibiotic-triggered IBS has been present for months or years, the microbiome disruption is more entrenched and the recovery takes longer. Six months of consistent daily synbiotic use is a realistic target for people with established post-antibiotic IBS. Improvement typically begins at the 4 to 8 week mark and continues building through the full protocol period.
The Treatment Approach That Addresses the Root Cause
Most IBS treatment is symptomatic — antispasmodics for cramping, loperamide for diarrhea, laxatives for constipation. These manage symptoms without touching the underlying microbiome disruption that’s causing them. For antibiotic-triggered IBS specifically, the microbiome is the root cause — and treating it directly produces more lasting improvement than symptom management alone.
A multi-strain synbiotic — the most important intervention. The breadth of strains matters more for antibiotic-triggered IBS than for general gut health — because the disruption affects multiple bacterial populations simultaneously and all of them need support. Seed DS-01 with its 24 clinically studied strains including multiple Bifidobacterium species — critical for motility regulation, butyrate production, and visceral sensitivity modulation — is the most comprehensively appropriate product for this specific context. Take it daily for a minimum of 6 months if IBS has been established for some time.
Address the root cause of antibiotic-triggered IBS
Seed DS-01 — 24 clinically studied strains targeting motility, gut lining integrity, visceral sensitivity, and bacterial balance. The most comprehensively appropriate synbiotic for post-antibiotic IBS. Commit to 6 months for established IBS.
👉 Check the current price of Seed DS-01 on Amazon
Digestive enzymes with every meal. Reducing the undigested food substrate that reaches dysbiotic bacteria reduces the fermentation-driven bloating and cramping that makes antibiotic-triggered IBS so symptomatic. Zenwise Digestive Enzymes taken at the start of every meal provides this support consistently. 👉 Check the price on Amazon.
Gut lining support through collagen. The increased permeability that underlies antibiotic-triggered IBS requires structural repair alongside bacterial repopulation. Multi collagen peptides daily provide the specific structural proteins the gut lining uses to rebuild tight junction integrity.
Anti-inflammatory curcumin. The chronic low-grade gut inflammation that perpetuates antibiotic-triggered IBS symptoms responds to curcumin’s NF-κB inhibition. Turmeric curcumin with black pepper taken daily reduces the inflammatory component that makes IBS symptoms more severe and persistent.
Fermented foods daily. Natural bacterial diversity from fermented foods accelerates microbiome rebuilding in ways that supplements alone don’t fully replicate. Kefir, yogurt, kimchi, and sauerkraut daily throughout the recovery period. The fermented foods that make the biggest impact on gut recovery gives you the practical list.
Soluble fiber — not insoluble. Psyllium husk normalizes IBS bowel patterns from both directions — constipation and diarrhea — through its gel-forming soluble fiber rather than bulk-adding insoluble fiber. Psyllium husk capsules daily are one of the most evidence-backed interventions for IBS bowel normalization regardless of type.
Frequently Asked Questions
Can antibiotics cause IBS?
Yes — post-infectious IBS following antibiotic use is a documented clinical phenomenon. Research consistently shows that antibiotic exposure, particularly broad-spectrum antibiotics and multiple courses, significantly increases the risk of developing IBS in the following months. The mechanism — microbiome disruption driving visceral hypersensitivity, altered motility, and increased gut permeability — is well understood.
How do I know if antibiotics triggered my IBS?
The clearest indicator is temporal — if IBS symptoms developed or significantly worsened in the weeks to months following an antibiotic course, post-infectious triggering is highly plausible. Additional indicators include: the IBS started after a specific antibiotic course you can identify, your gut was normal before that course, and your symptoms fit the pattern of dysbiosis-driven IBS rather than primarily genetic or stress-driven patterns.
Can antibiotic-triggered IBS be cured?
Cured is a strong word — IBS treatment is about management and substantial improvement rather than elimination for most people. However, antibiotic-triggered IBS responds better to targeted microbiome intervention than IBS without an identifiable bacterial trigger — because the root cause is more specifically addressable. Many people with post-antibiotic IBS achieve very significant symptom reduction with 6 months of consistent synbiotic use combined with the dietary and supplement approaches above.
What is the best probiotic for IBS caused by antibiotics?
A multi-strain synbiotic with multiple Bifidobacterium and Lactobacillus strains, acid-resistant delivery, and prebiotic included. Seed DS-01 is our top recommendation. Read our full Seed DS-01 review here. 👉 Check the price on Amazon.
How long does post-antibiotic IBS last?
Without active intervention — potentially years, with some people never fully recovering to their pre-antibiotic baseline. With consistent active microbiome support — 6 months of daily synbiotic use, fermented foods, prebiotic fiber, and gut lining support — most people with post-antibiotic IBS see significant improvement. The longer the IBS has been established, the longer recovery takes.
Does post-antibiotic IBS go away on its own?
For some people with mild disruption — yes, over many months. For people with significant disruption or multiple antibiotic courses — passive waiting produces incomplete recovery at best. Signs your gut still hasn’t recovered helps you identify whether passive recovery is working or whether active intervention is needed.
Your IBS Has a Name — and a Cause — and a Path Forward
There’s something both validating and empowering about understanding that your IBS has a specific, identifiable trigger. It’s not arbitrary. It’s not permanent. It’s not just how your gut is now and forever. It’s a functional disruption with a known mechanism that responds to targeted intervention.
The antibiotics disrupted your microbiome. The microbiome disruption produced the gut dysfunction driving your IBS symptoms. Restoring the microbiome — deliberately, consistently, over sufficient time — addresses the root cause in a way that symptom management alone never can.
Start the protocol. Give it six months of genuine consistency. Your gut was different before — and with the right support, it can be different again.
Your post-antibiotic IBS recovery stack
👉 Seed DS-01 Daily Synbiotic — the probiotic foundation, 6 months minimum
👉 Zenwise Digestive Enzymes — reduce bloating and cramping with every meal
👉 Psyllium Husk Capsules — normalize IBS bowel patterns
👉 Multi Collagen Peptides — repair gut lining permeability
More from TummyCure:
- How Long Does Your Gut Take to Recover After Antibiotics?
- The Best Probiotic to Take After Antibiotics
- Signs Your Gut Still Hasn’t Recovered After Antibiotics
- Your Gut Recovery Grocery List
- Best Probiotic for IBS-C, IBS-D and IBS-M
- Do Digestive Enzymes Help IBS?
- Can Leaky Gut Cause Anxiety and Depression?
- Seed DS-01 — Full Review
- How Antibiotics Can Wreck Your Gut for a Year
- The Complete Gut Health Guide
About the Author
Rachel Donnelly is a certified nutritional health coach and gut health writer who spent years struggling with IBS and bloating before making digestive wellness her specialty. She writes for TummyCure with one goal: cut through the noise and tell you what actually works. When she’s not deep in microbiome research, she’s fermenting things in her kitchen and losing arguments with her husband about whether kombucha counts as a dessert.
As an Amazon Associate we earn from qualifying purchases through some links in our articles.