If you’re doing everything right and still can’t lose weight, your gut microbiome may be the missing variable. Here’s the science behind how gut bacteria control weight — and what to do about it.
You’re Doing Everything Right — So Why Isn’t the Scale Moving?
You’ve cut calories. You’re exercising more. You’re tracking what you eat. And the weight either won’t come off, comes off agonizingly slowly, or comes right back the moment you ease up. You’ve started to wonder if there’s something fundamentally wrong with your metabolism — or worse, if you’re just not trying hard enough.
There’s a third possibility that gets almost no attention in mainstream weight loss conversations — and it’s one that the research is increasingly pointing toward as a genuinely significant factor for a large proportion of people who struggle with their weight: your gut microbiome.
The bacteria living in your intestines aren’t passive passengers. They actively regulate how calories are extracted from food, how your hunger and fullness hormones function, how your metabolism operates, and how your body responds to insulin. Two people can eat identical diets and exercise identical amounts and have meaningfully different weight outcomes — and the gut microbiome is increasingly understood to be one of the most significant variables explaining why.
Start addressing your gut-weight connection today
Seed DS-01 Daily Synbiotic — 24 clinically studied strains targeting microbiome diversity, metabolic function, and gut health. The foundation of any gut-based approach to weight management.
The Research That Changed How Scientists Think About Weight
The gut-weight connection moved from fringe theory to mainstream science largely on the back of a landmark series of experiments involving germ-free mice — mice raised in sterile environments with no gut bacteria whatsoever.
Researchers found that germ-free mice were dramatically leaner than normal mice — even when fed the same diet. When gut bacteria from obese mice were transplanted into germ-free mice, those mice gained significantly more fat than mice that received bacteria from lean donors — eating the same food in the same amounts. The bacteria themselves were driving different metabolic and fat storage outcomes from identical inputs.
The human data followed. A 2013 study transplanting gut bacteria from obese human twins into germ-free mice produced obese mice. Bacteria from lean twins produced lean mice. Same food. Same conditions. Different bacteria — different body composition.
These experiments didn’t prove that gut bacteria are the only determinant of weight — they’re not. But they established beyond reasonable scientific doubt that the gut microbiome is a meaningful and previously underappreciated variable in weight regulation — one that interacts with diet and exercise rather than replacing them as explanations.
How Your Gut Bacteria Affect Your Weight — The Specific Mechanisms
Understanding the specific ways gut bacteria influence weight makes the connection tangible rather than abstract — and points toward what actually changes it.
Calorie Extraction From Food
Your gut bacteria determine how many calories you actually absorb from the food you eat — and this varies more significantly between individuals than most people realize. Certain bacterial species are highly efficient at extracting energy from carbohydrates and fiber that your own digestive enzymes can’t break down. People whose gut microbiome is dominated by these highly efficient extractors absorb more calories from the same food than people with different bacterial compositions.
This means that calorie counting — already an imprecise science — is further complicated by the fact that your gut bacteria determine what percentage of those calories you actually absorb. Two people eating 2,000 calories may be absorbing meaningfully different amounts based purely on their microbiome composition.
Hunger and Satiety Hormones
Your gut bacteria communicate directly with the cells that produce your hunger and fullness hormones — particularly GLP-1 (yes, the same hormone that Ozempic and Wegovy mimic), PYY, and ghrelin. Certain beneficial bacterial species — particularly Akkermansia muciniphila and specific Bifidobacterium strains — stimulate the natural production of GLP-1 and PYY, which suppress appetite and increase satiety. Dysbiotic microbiomes produce less of these natural appetite-suppressing signals and more of the hunger-promoting signals that drive overeating.
This means that a disrupted gut microbiome can produce genuine biological hunger signals that override willpower and discipline — not a character flaw but a hormonal consequence of bacterial imbalance. The person who feels constantly hungry despite eating what should be enough isn’t lacking self-control — their gut bacteria are producing hormonal signals that make satiety harder to achieve.
Insulin Sensitivity and Blood Sugar Regulation
The gut microbiome plays a central role in insulin sensitivity — how efficiently your cells respond to insulin and clear glucose from the bloodstream. Specific bacterial metabolites, particularly short-chain fatty acids produced by butyrate-producing species like Faecalibacterium prausnitzii and Roseburia, improve insulin sensitivity through their effects on gut hormone signaling and systemic inflammation.
When these butyrate-producing species are depleted — which happens through poor diet, antibiotic exposure, and chronic stress — insulin sensitivity declines. Poor insulin sensitivity means blood sugar spikes higher after meals, insulin is secreted in larger amounts, and more glucose gets stored as fat rather than used for energy. The result is metabolic dysfunction that makes weight loss harder and weight gain easier — driven at its root by bacterial depletion.
Gut Inflammation and Metabolic Dysfunction
Certain gut bacterial species — particularly gram-negative bacteria whose cell walls contain lipopolysaccharide (LPS) — produce compounds that, when they cross the gut barrier into the bloodstream, trigger systemic inflammation. This metabolic endotoxemia — chronic low-grade inflammation from gut bacterial products in the circulation — is consistently associated with obesity, insulin resistance, and metabolic syndrome in research.
The connection goes both ways — obesity disrupts the gut microbiome, and a disrupted microbiome promotes the metabolic dysfunction that drives obesity. Breaking this cycle requires addressing the microbiome directly rather than only focusing on diet and exercise.
This is also why gut permeability affects so much more than just digestion — the inflammatory compounds crossing a leaky gut barrier contribute to the systemic metabolic dysfunction that makes weight management harder.
Fat Storage Regulation
Gut bacteria influence the expression of genes involved in fat storage — particularly FIAF (fasting-induced adipose factor), which regulates fat storage in adipose tissue. Certain bacterial species suppress FIAF expression, promoting greater fat storage from the same caloric intake. When these species dominate the microbiome — as they do in dysbiotic, low-diversity gut environments — fat storage is promoted at the genetic expression level regardless of calorie intake.
This mechanism was one of the original findings in the germ-free mouse experiments — germ-free mice had higher FIAF expression and stored less fat than normal mice, even eating more food.
The Microbiome Composition of Lean vs Overweight People
Research consistently finds measurable differences in gut microbiome composition between lean and overweight individuals — across cultures, diets, and geographic locations. The pattern that emerges is consistent enough to be genuinely meaningful.
Lean individuals tend to have higher overall microbiome diversity, higher proportions of Bacteroidetes relative to Firmicutes, higher levels of butyrate-producing species like Faecalibacterium prausnitzii, higher levels of Akkermansia muciniphila — a species associated with metabolic health and gut lining integrity — and lower levels of inflammatory LPS-producing species.
Overweight individuals tend to show the opposite pattern — lower diversity, higher Firmicutes relative to Bacteroidetes, depleted butyrate producers, lower Akkermansia, and higher inflammatory species. These differences aren’t just correlational — the transplant experiments demonstrate they’re causally related to metabolic outcomes.
What Disrupts the Gut Bacteria That Regulate Weight
Understanding what damages the weight-regulating microbiome helps identify what needs to change alongside probiotic support.
Ultra-processed food and added sugar are the most significant dietary drivers of metabolic dysbiosis. They feed Firmicutes and harmful species while starving the beneficial bacteria that regulate hunger hormones and insulin sensitivity. The Western diet — high in processed food, low in fiber — consistently produces the dysbiotic microbiome pattern associated with weight gain. The foods that damage gut bacterial balance are largely the same ones that promote weight gain — not coincidentally.
Antibiotics — each course reduces microbiome diversity and depletes beneficial species including butyrate producers and Akkermansia. Multiple antibiotic courses are consistently associated with higher BMI in population studies. If you’ve taken multiple antibiotic courses over the years and have struggled with weight since, the connection is worth taking seriously. How long gut recovery after antibiotics actually takes explains why the metabolic effects can persist long after the course is finished.
Chronic stress shifts the microbiome toward dysbiotic compositions through cortisol’s effects on gut barrier function and bacterial populations. Stress also drives the cortisol-insulin axis in ways that promote abdominal fat storage independently — making chronic stress a double metabolic problem that includes but goes beyond the gut.
Poor sleep disrupts the circadian regulation of both metabolism and gut bacteria simultaneously. Short or irregular sleep is consistently associated with gut microbiome changes that resemble the dysbiotic pattern seen in overweight individuals — independent of diet.
Sedentary lifestyle reduces gut microbiome diversity directly. Regular exercise consistently increases beneficial bacterial populations and microbiome diversity in research — making movement a gut health intervention as much as a metabolic one.
Can Fixing Your Gut Help You Lose Weight?
This is the most practically important question — and the honest answer is nuanced.
Improving your gut microbiome is not a weight loss strategy on its own. It doesn’t replace calorie awareness, food quality, or physical activity. But for people whose weight management has been frustratingly resistant to standard approaches, addressing the microbiome may be the variable that makes those standard approaches actually work.
Research on probiotic supplementation and weight shows modest but consistent effects — reductions in body weight, BMI, and waist circumference in overweight subjects using multi-strain probiotic supplements over 8 to 12 weeks. The effect sizes are modest — typically 1 to 2 kg of additional weight loss compared to placebo — but these are additive effects on top of whatever other approach is being used, not standalone results.
More significantly, probiotic supplementation consistently improves the metabolic markers underlying weight management difficulty — insulin sensitivity, inflammatory markers, hunger hormone levels, and gut barrier integrity. These improvements create a biological environment in which weight loss is easier to achieve and maintain.
The most comprehensive view of how gut health fits into weight management — and how all the pieces connect — is in The Complete Gut Health Guide.
The Dietary Approach That Supports Weight-Regulating Gut Bacteria
High fiber from diverse plant sources is the single most impactful dietary change for improving the weight-regulating microbiome. Fiber feeds the butyrate-producing species, Akkermansia, and other beneficial bacteria that improve insulin sensitivity and hunger hormone function. The research is clear that dietary fiber diversity — eating many different plant foods rather than high amounts of a few — produces the greatest microbiome diversity gains.
Aim for 30 or more different plant foods per week — vegetables, fruits, legumes, whole grains, nuts, seeds, and herbs all count. This isn’t as difficult as it sounds — a varied diet naturally gets there when you prioritize plant food diversity over consistency.
Fermented foods daily increase Akkermansia and beneficial bacterial diversity in ways that supplement alone don’t fully replicate. The fermented foods that make the biggest difference — kefir, yogurt, kimchi, sauerkraut — provide natural bacterial diversity alongside their metabolic benefits.
Reducing ultra-processed food and added sugar removes the primary dietary inputs that shift the microbiome toward the dysbiotic, high-Firmicutes pattern associated with weight gain and metabolic dysfunction.
Adequate protein supports the lean mass preservation that maintains metabolic rate during weight loss — and certain protein sources, particularly fermented dairy and legumes, provide additional microbiome benefits alongside the macronutrient value.
For consistent daily prebiotic fiber support — particularly useful when dietary fiber intake is variable — psyllium husk provides reliable soluble prebiotic fiber that feeds beneficial bacteria consistently.
Daily prebiotic fiber — feeding your weight-regulating bacteria
Psyllium Husk Capsules — the most research-backed fiber supplement available. Consistent daily prebiotic support for the beneficial bacteria that regulate hunger hormones and insulin sensitivity. Two capsules with a large glass of water daily.
👉 Check the current price of Psyllium Husk Capsules on Amazon
The Probiotic Approach — What the Research Supports
For directly addressing the microbiome composition associated with weight management difficulty, a quality multi-strain synbiotic is the most evidence-backed supplementation approach.
The strains most relevant to metabolic outcomes include Bifidobacterium species — which improve insulin sensitivity and reduce inflammatory markers — and Lactobacillus strains that have been specifically studied for effects on body composition and metabolic health. A product covering multiple strains from both genera provides the broadest metabolic microbiome support.
Seed DS-01 includes 24 clinically studied strains with specific research behind each one — including strains studied for cardiovascular and metabolic health markers alongside the core digestive and immune benefits. It’s the most comprehensively researched synbiotic available and the most appropriate choice for someone approaching weight management from a gut health angle.
Address the gut bacteria driving your weight management difficulty
Seed DS-01 — 24 clinically studied strains including strains researched for metabolic health, insulin sensitivity, and gut barrier function. The most comprehensive synbiotic for the gut-weight connection. Commit to 90 days of consistent daily use.
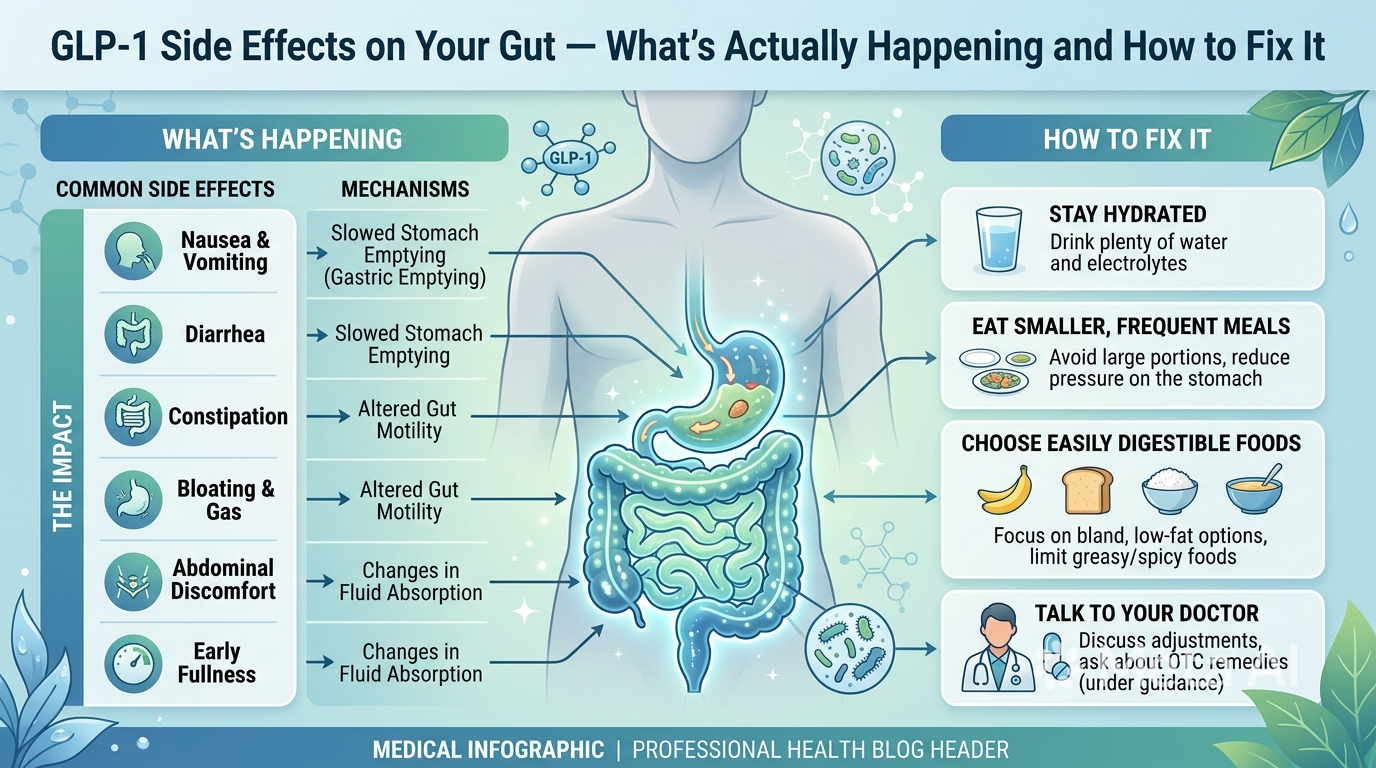
The GLP-1 Connection — Why Gut Bacteria and Weight Loss Medications Work Together
If you’re on a GLP-1 medication like Ozempic or Wegovy — or considering one — the gut-weight connection is particularly relevant. GLP-1 medications work by mimicking a hormone that your gut bacteria help produce naturally. A healthier microbiome may actually enhance your response to GLP-1 medications through its effects on natural GLP-1 receptor sensitivity.
Additionally, the gut microbiome changes that GLP-1 medications produce — through altered transit and reduced food intake — need active management to ensure the metabolic benefits of better bacterial composition don’t get offset by dysbiotic disruption from the medication itself. Whether to take a probiotic on GLP-1 medications covers this connection in detail.
Frequently Asked Questions
Can gut bacteria really affect weight loss?
Yes — through multiple specific mechanisms including calorie extraction efficiency, hunger and satiety hormone regulation, insulin sensitivity, fat storage gene expression, and systemic inflammation. The research is robust enough that the gut microbiome is now considered a meaningful variable in weight regulation, not just a correlate of weight status.
What gut bacteria help with weight loss?
Akkermansia muciniphila is the most researched species for metabolic health and weight regulation. Butyrate-producing species like Faecalibacterium prausnitzii improve insulin sensitivity. High-Bacteroidetes relative to Firmicutes ratios are consistently associated with leaner body composition. High overall microbiome diversity is the most consistent predictor of metabolic health across populations.
Can probiotics help you lose weight?
Research shows modest but consistent effects — 1 to 2 kg of additional weight loss over 8 to 12 weeks compared to placebo in overweight subjects. More significantly, probiotics improve the underlying metabolic markers — insulin sensitivity, inflammatory markers, hunger hormone levels — that determine how easily weight loss is achieved and maintained.
What is the best probiotic for weight loss?
A multi-strain synbiotic with multiple Bifidobacterium strains — which have the most research for metabolic outcomes — alongside Lactobacillus strains studied for body composition effects. Seed DS-01 is our top recommendation. Read our full Seed DS-01 review here.
Does gut health affect belly fat specifically?
Yes — abdominal fat is particularly associated with the systemic inflammation and insulin resistance that dysbiotic gut bacteria promote. Improving gut microbiome composition consistently reduces inflammatory markers and improves insulin sensitivity — both of which are specifically associated with visceral abdominal fat reduction.
How long does it take for gut bacteria changes to affect weight?
Meaningful microbiome composition shifts take 8 to 12 weeks of consistent dietary changes and probiotic supplementation. Metabolic improvements — in insulin sensitivity, hunger hormone levels, inflammatory markers — become measurable in this window. Scale-based weight changes attributable to microbiome improvement appear over 3 to 6 months of sustained intervention.
Your Gut Bacteria Aren’t Working Against You — They Just Need the Right Support
The frustration of doing everything right and still not seeing results is real — and for a significant number of people, the gut microbiome is a genuine contributing factor to that frustration. Not the only factor. Not a magic solution. But a real, specific, addressable variable that current mainstream weight loss advice almost completely ignores.
Improve the microbiome and you improve the biological environment in which weight loss happens. Better hunger hormone signaling. Better insulin sensitivity. Less inflammatory drive toward fat storage. More efficient metabolic function overall. These changes don’t happen overnight — they build over 3 to 6 months of consistent dietary and probiotic support.
But they’re real. And for people who have been struggling with weight despite genuine effort, addressing the gut is often the variable that finally makes the effort pay off.
Start addressing the gut-weight connection today
👉 Seed DS-01 Daily Synbiotic — the microbiome foundation
👉 Psyllium Husk Capsules — daily prebiotic fiber for metabolic bacteria
👉 Zenwise Digestive Enzymes — optimize food breakdown alongside microbiome work
👉 Turmeric Curcumin Gummies — reduce the gut inflammation driving metabolic dysfunction
More from TummyCure:
- Is Your Gut the Reason You Can’t Lose Weight?
- Seed DS-01 — Full Review
- Should You Take a Probiotic on GLP-1 Medications?
- Complete GLP-1 Gut Side Effects Guide
- Can Leaky Gut Cause Anxiety and Depression?
- Foods That Cause Leaky Gut
- 10 Fermented Foods That Changed My Digestion
- 8 Signs Your Gut Desperately Needs a Probiotic
- The Complete Gut Health Guide
- Probiotic vs Synbiotic — What’s the Difference?
About the Author
Rachel Donnelly is a certified nutritional health coach and gut health writer who spent years struggling with IBS and bloating before making digestive wellness her specialty. She writes for TummyCure with one goal: cut through the noise and tell you what actually works. When she’s not deep in microbiome research, she’s fermenting things in her kitchen and losing arguments with her husband about whether kombucha counts as a dessert.
As an Amazon Associate we earn from qualifying purchases through some links in our articles.